Your cart is currently empty!
Tag: Dumfries and Galloway

Operation of pass box transferring of materials in Microbial lab of Pharma Industry
Operation of pass box. transferring of materials in Microbial lab of Pharma Industry To lay down the pass-box operation procedure. This procedure is applicable when passing the material from one room to another using the pass-box. PROCEDURE: General cleaning After use, wipe the inside of the folded-box clean dry rag. After completing the mechanical cleaning…

Safety working in the microbiological laboratory In Pharma
Safety working in the microbiological laboratory In Pharma Providing a safe work environment to employees. The personnel of the microbiological laboratory. PROCEDURE: To work in the laboratory allowed a person for a recent preliminary medical examination, an introduction, and initial instruction in the workplace on occupational safety, special education, if necessary (including safety), and validation…

Requirements to personnel of the miсrobiological laboratory
Requirements for personnel of the miсrobiological laboratory Set requirements to personnel of the microbiological laboratory Determine the qualifications of personnel, their rights and responsibilities, and also official relationships. PROCEDURE Requirements, rights, and duties of employees of the microbiological laboratory are determined in duty regulations. On arrival at work, the employee must be acquainted with corresponding…

Manufacturing Operations and Controls In Pharma Industry
Manufacturing Operations and Controls In Pharma Industry All manufacturing operations shall be carried out under the supervision of technical staff approved by the Licensing Authority. Each critical step in the process relating to the selection, weighing, and measuring of raw material addition during various stages shall be performed by trained personnel under the direct personal…

Disposal of waste
Disposal of waste Disposal of waste means eliminating, scraping, recycling, or destroying unwanted materials known as waste that is generated from the manufacturing, packing, and warehousing of industrial pharmaceutical products. Following the correct methods for waste disposal will ensure lesser pollution and hazards to the environment. All the pharmaceutical waste shall be collected initially in…

Handling of external laboratory activities
Handling of external laboratory activities To lay down a procedure for qualifying, auditing, sending samples with reference to testing procedures, and obtaining the results for the same. This SOP is applicable to all the external laboratories to which the samples are sent for testing due to the non-availability of in-house facilities/resources. PROCEDURE The external laboratory…

Procedure of laundry
Procedure of laundry To lay down the procedure for describing the steps to be followed for laundry the uniform. This Procedure is applicable to laundry. PROCEDURE Uniforms should be transferred from the production area in special closed plastic bags separately for each section like Packing, core area, and Warehouse. Clothes should be washed in accordance…

EXCEL SHEET VALIDATION
EXCEL SHEET VALIDATION The objective of this procedure is to establish the guideline for the validation of Microsoft Excel spreadsheets use for calculating the product quality parameters. This procedure covers the validation procedure for validation of Microsoft Excel spreadsheets use for calculating the product quality parameters in QA. Procedure Spreadsheets used for simple calculations (e.g:…

CALIBRATION OF DIES REQUIRED FOR BLEND SAMPLING
CALIBRATION OF DIES REQUIRED FOR BLEND SAMPLING The objective of this procedure is to establish the guideline for the calibration of dies required for blend sampling. This SOP is applicable to dies required for a sampling of the blend for validation batches manufactured in the Pharma Industry. Procedure Take a die to be calibrated for…

HANDLING COUNTERFEIT/FALSIFIED MATERIALS
HANDLING COUNTERFEIT/FALSIFIED MATERIALS To lay down a standard procedure that describes handling counterfeit/falsified materials. Applicable for the procedure of handling counterfeit/falsified materials. PROCEDURE The received material should undergo incoming inspection. If the incoming inspection results of the materials are unsatisfactory, the QA department initiates the investigation of the case. The Authorized person prepares a report…

Technology Transfer
Technology Transfer To lay down a procedure for technology transfer of new products to be taken. The scope of this procedure is to all the new Products for which Technology has been provided by an outside party PROCEDURE Whenever any NEW PRODUCT is to be taken at Pharmaceutical Industry & the technology has been provided…

Shipping Validation
Shipping Validation Objective The objective of this study is to establish a procedure to record temperature data to ensure that transportation conditions have not adversely affected a product until it reaches the Central Warehouse when transported as per regular Shipment practice. Scope The protocol shall be applicable at the formulation Plant. The Shipping (Transportation) validation…

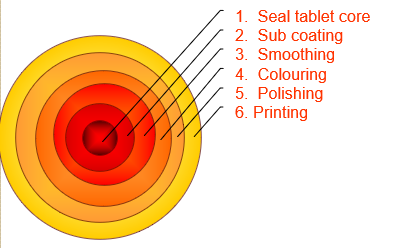
COATING PANS
COATING PANS CONTENTS: COATING PROCESSES TYPES OF COATING PANS CONVENTIONAL COATING PANS PERFORATED COATING PANS VALIDATION OF COATING PANS IQ OQ PQ REFERENCES Main coating processes Film coating Sugar coating Sugar coating Traditionally sugar coatings formed the bulk of coated tablets but today film coatings are the more modern technology in tablet coating. Description of…

AUTOCLAVE – MAINTENANCE AND VALIDATION
AUTOCLAVE – MAINTENANCE AND VALIDATION. INTRODUCTION An autoclave is a device to sterilize equipment and supplies by subjecting them to high pressure steam at 121° C or more. It was invented by Charles Chamberland in 1879,although a precursor known as the steam digester was created by Denis Papin in 1679. The term autoclave is also…

WEIGHING ON AN ANALYTICAL BALANCE AS PER USP (1251)
WEIGHTS AND BALANCES AND ANALYTICAL BALANCE The requirements for weights into conformity with American National Standard ANSI/ASTM E617, “Laboratory Weights and Precision Mass Standards.” Pharmacopeial tests and assays require balances that vary in capacity, sensitivity, and reproducibility. Unless otherwise specified, when substances are to be “accurately weighed” for Assay, the weighing is to be performed…

Pharma Job WhatsApp group link
Pharma Job WhatsApp group link pharma WhatsApp group link Pharma Jobs (90) https://chat.whatsapp.com/Fw14tT8ThZF0pV0pMGDVZ9 Pharma Jobs Hub 04 (116) https://chat.whatsapp.com/Fc0f8DpBucQ5jc7ltXe1Uy Pharma Jobs Hub 03 (82) https://chat.whatsapp.com/HohCLKKqLU34chFxjg2GGW Pharma Jobs Hub 02 (93) https://chat.whatsapp.com/DaH99Qu1Ato9NmBx4tzVo5 Pharma Jobs and Post (219) https://chat.whatsapp.com/Dy1GkgHZloYJPsfzIC6tnZ Pharma Jobs and Post 02 (196) https://chat.whatsapp.com/La8yGcVqeknKkfEeymYwMv Pharma Jobs and Post 03 (157) https://chat.whatsapp.com/CLI4oqKyVAf9nKNbbOz1Hc Pharma Jobs and Post04…

Pharma Definitions
Pharma Definitions 1. Aseptic processing A process performed maintaining the sterility of a product that is assembled from components, each of which has been sterilized by steam, dry heat, ionizing radiation, gas or sterile filtration. This is achieved by using conditions and facilities designed to prevent microbiological contaminants. 2. Bioburden The total number of micro-organisms…
Dispensing of raw material
Dispensing of raw material OBJECTIVE Procedure of dispensing raw materials. SCOPE This SOP is applicable for dispensing of all raw materials. RESPONSIBILITY Warehouse, production and QA staff shall be responsible to follow the procedure mentioned in this SOP. ACCOUNTABILITY Warehouse In-Charge and QA Head shall be accountable for compliance of the procedure mentioned in this…
Operation and cleaning of vacuum cleaner
Operation and cleaning of vacuum cleaner OBJECTIVE Procedure for operation and cleaning of vacuum cleaner. SCOPE This procedure is applicable in the Warehouse RESPONSIBILITY Warehouse personnel shall be responsible to follow the procedure mentioned in this SOP. ACCOUNTABILITY Warehouse In-Charge and QA Head shall be accountable for compliance of the procedure mentioned in this SOP.…
Temperature and humidity monitoring
Temperature and humidity monitoring OBJECTIVE The procedure of temperature and humidity monitoring in the storage and dispensing area. SCOPE This SOP is applicable for monitoring temperature and humidity in the storage area/ dispensing area. RESPONSIBILITY Warehouse personnel shall be responsible to follow the procedure mentioned in this SOP. ACCOUNTABILITY Warehouse In-Charge and QA Head shall…